A Case Study: Control Strategies For A Multi-Dose Pen Injector
By Steven Poon, principal consultant, 7TP Consultancy

In part 1 of this series, we discussed defining combination product critical quality attributes (CQAs) and the importance of having a control strategy with risk management considerations. Here, we will discuss a case study highlighting the process of defining a control strategy for assessing combination products’ CQAs.
Similar to risk management, the control strategy is a living process that must be kept up to date alongside the combination product development and post-production activities. Figure 1 provides an overview of the control strategy process, implementing the steps outlined in part 1.
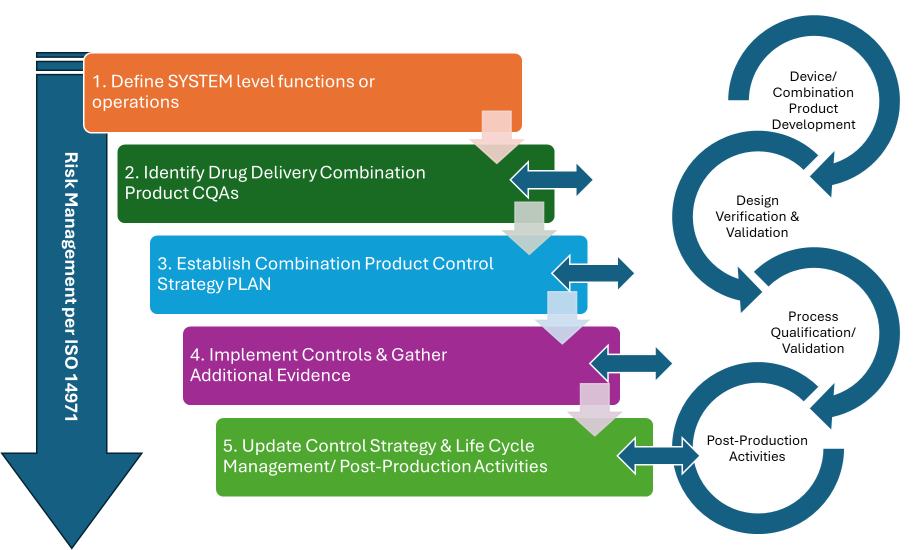
Figure 1: Overview of the control Strategy process
To illustrate the steps with a few examples, we will discuss a multi-dose pen injector-based drug delivery combination product as a case study.
1. Define SYSTEM Level Functions Or Operations
A typical multi-dose pen injector-based drug delivery combination product would consist of the following SYSTEM level functions/operations (Table 1):
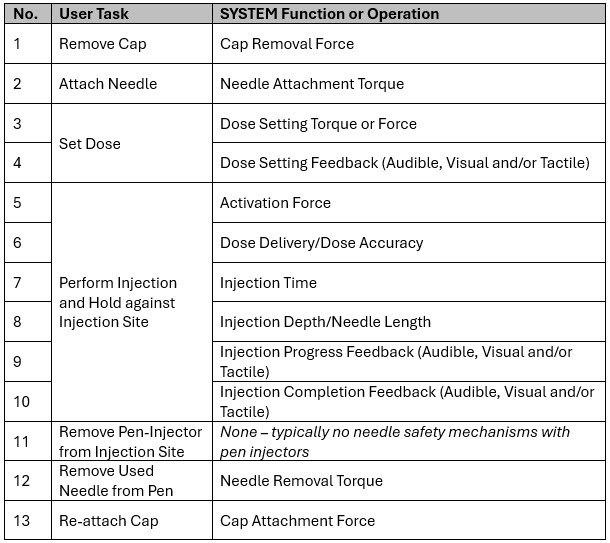
Table 1: Example of pen injector-based combination product SYSTEM level functions or operations.
Although there may be other functions/operations of the product – such as break-loose and glide force of the cartridge, or spring forces – they would be considered as lower-level or sub-level function/operation. It is important to focus on what the finished product does and how the user interacts with the product to define the SYSTEM. Subsystem functions come into play when establishing the control strategy plan.
2. Identify Combination Product CQAs
With your list of SYSTEM functions or operations defined, using your risk-based approach, the next step is to identify the relevant combination product CQAs. An example of how this can be done (without going into details of how to identify PFs and EDDOs, as these are already covered by their respective references1,2) is shown in Table 2.
Note that some of these responses, and therefore identification as a combination product CQA, will depend on the drug product, intended use, and indications/disease state being treated. For this example, this drug product is for non-emergency use, where failure to immediately dose is not life-threatening and would not lead to a catastrophic harm.
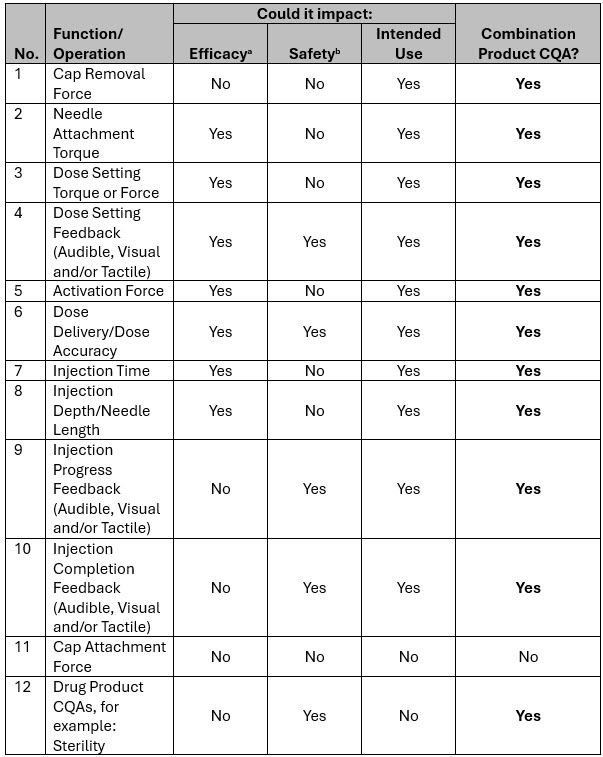
Table 2: Example of pen-Injector-based combination product CQAs. a. Efficacy means accurate dose delivery to the intended injection site. b. Safety means freedom from unacceptable harm, where harm considers any end effect of serious or more. This also considers harm to any user, as opposed to direct harm to the patient per definition of primary function.
Table 2 does not provide an exhaustive list of drug product CQAs, but where drug product CQAs could also impact efficacy, safety, or intended use, they should also be assessed and designated as combination product CQAs.
3. Establish Control Strategy Plan
With your list of combination product CQAs defined, they next should be mapped against the various control steps (Figure 2, Part 1) to identify your known-knowns and known-unknowns. Table 3 provides a sample of combination product CQAs and the potential defined controls.
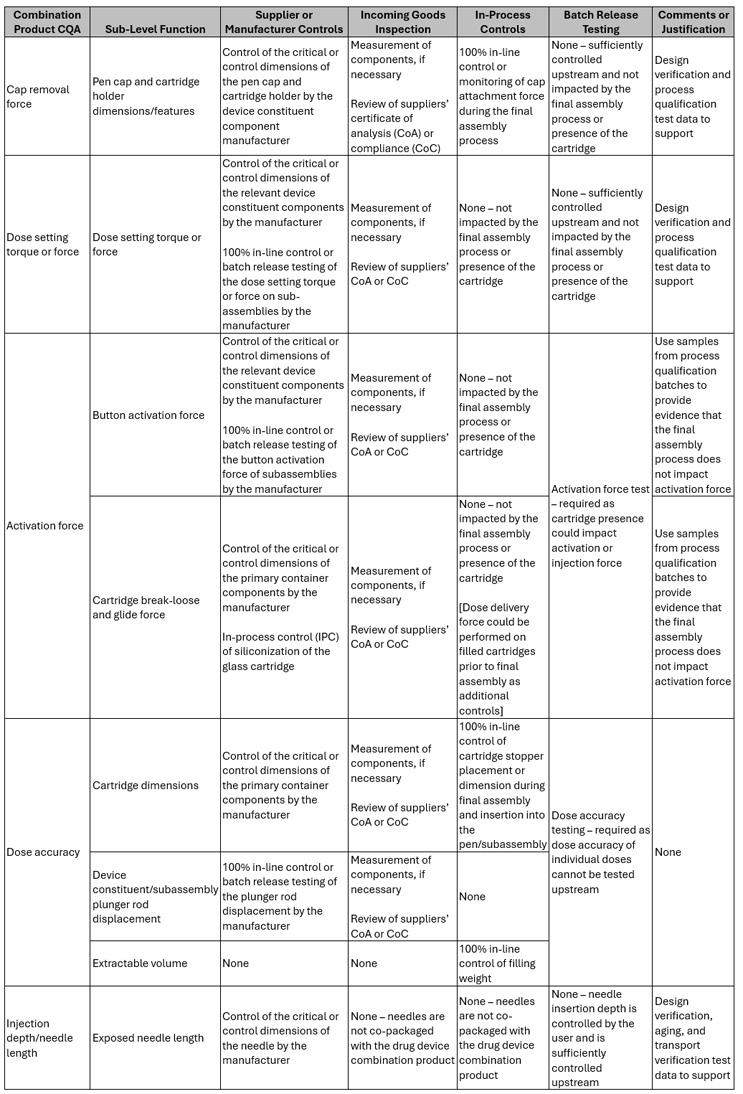
Table 3: A sample of combination product CQAs and relevant upstream and downstream controls.
4. Implement Controls And Gather Additional Evidence
Controls as identified in your control strategy plan should be implemented and verified for their effectiveness. Where there is justification for not having controls, evidence of this should also be provided through testing of batches from design verification, process qualification, transport verification, or other stress conditions that were deemed necessary through risk management.
5. Post-Production Activities
The data collected during development and from post-production activities should provide feedback to the control strategy. This analysis could indicate additional controls may be needed in some areas or that sample sizes can be adjusted based on successful batch production.
Conclusion
As the emergence of combination products continues to grow, and as the industry moves toward novel and platform products, the historic walls between primary packaging, device development, industrialization, and drug product/analytical teams need to be broken down. Early engagement between drug product and device functions is key to having a holistic and end-to-end view of what needs to be controlled and the controls that can and should be implemented. Teams should align on definitions early in development, understand that there will some differences in approaches, and be open to learnings from both sides – be pragmatic. Keeping in mind the ultimate destination remains the same: to have safe and effective products that can be used by those who need them.
About The Author:
 Steven Poon has been working in the medical device and drug-delivery space for over 15 years, with former roles in product development consultancy, OEM medical device manufacturer, and biopharmaceuticals. He often acts as a bridge between medical device development and drug product teams. Once described by a client as a “Swiss army knife”, Steven combines a background in mechanical engineering, a keen eye for quality, and a natural interest in technical and regulatory developments.
Steven Poon has been working in the medical device and drug-delivery space for over 15 years, with former roles in product development consultancy, OEM medical device manufacturer, and biopharmaceuticals. He often acts as a bridge between medical device development and drug product teams. Once described by a client as a “Swiss army knife”, Steven combines a background in mechanical engineering, a keen eye for quality, and a natural interest in technical and regulatory developments.
