Closing The Adherence Gap: The Connected Health Era
By Ajay Chintawar, associate director, Teva Pharmaceuticals

For decades, a deep and invisible gap existed in modern treatment through injectables. A physician could prescribe a lifesaving drug, a pharmacist could dispense it, and a patient could walk home with it, but the moment that patient closed their front door, the treatment entered a secret phase. Did they take it? Did they take it correctly? Was it stored at the right temperature? For a long time, the only way to answer these questions was through a patient’s memory, feedback form, or a doctor’s guesswork during a follow-up months later.
This adherence gap is more than a logistical pain point; it is a clinical crisis. In chronic disease management, medication adherence to traditional injectables floats around just 50%. When dealing with complex therapies for multiple sclerosis, arthritis, or growth disorders, a few missed doses can trigger a relapse or permanent disability. This reality served as the primary trigger for the integration of connected health. We didn't just need better drugs; we needed a way to ensure those drugs actually made it into the patient's system.
Defining The Connected Ecosystem
Connected health is often misunderstood as simply adding Bluetooth to a gadget. In reality, it is a socio-technical model that creates a closed-loop feedback system between the patient, the device, and the healthcare professional. In the world of injectables, this means transforming a mechanical pen or an autoinjector into a data-capturing hub.
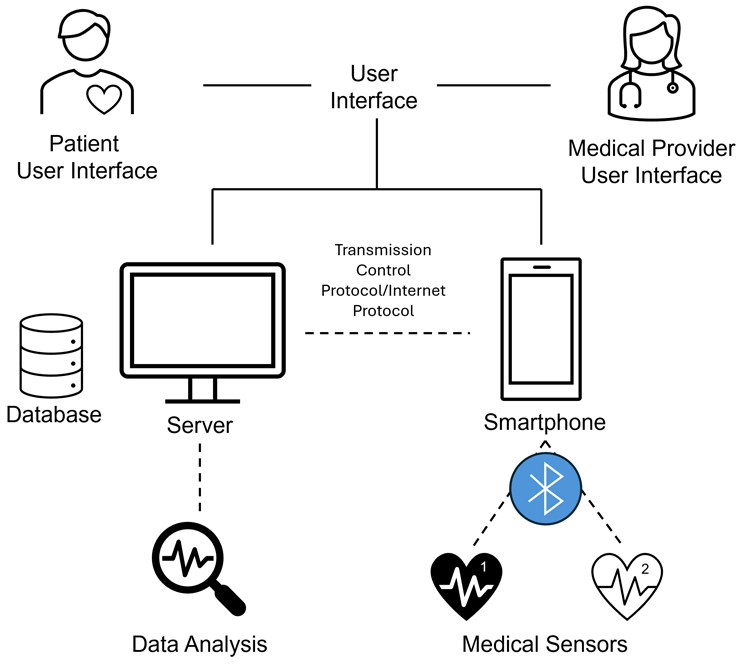
Figure 1. The flow of data from patient to cloud to doctor. Adapted from Scagliusi et al (CC BY 4.0). 1
The ecosystem typically consists of three layers:
- the hardware (a smart injector),
- the patient interface (a mobile app),
- the clinical dashboard.
By digitizing the injection event, we move from a reactive model to a proactive one, where a nurse can call a patient because the data shows they’ve missed consecutive doses.
We often forget that living with chronic conditions is a full-time job. For a patient with multiple sclerosis or type 1 diabetes, an injection isn’t just a medical task; it is a constant cognitive burden. Connected health addresses this mental load through digitally solving the responsibility crisis. By shifting the responsibility of remembering schedules from the human brain to the device, we reduce the anxiety and needle phobia that often lead to skipped doses.
A gentle vibration from a smart pen or a context-aware notification on a phone, reminding a patient to inject before they leave for work, with dosage history data at their fingertips, transforms a stressful chore into a seamless habit. This shift from being a passive recipient of care to an active partner who can see their own progress on a graph is psychologically empowering. It changes the internal narrative from "I am sick" to "I am in control of my data and my health."
The COVID-19 Catalyst: A Forced Evolution
While the move toward digital health was already boiling, the COVID-19 pandemic pushed it into overdrive. Almost overnight, remote monitoring went from a nice-to-have option to a necessity for survival. Before the crisis, many saw these tools as a futuristic luxury, but when hospitals closed their doors, digital tracking became the only way for patients to stay safe. This period proved once and for all that connected health isn't just a backup plan, it’s a smarter, more reliable way to handle medicine and it’s here to stay.
Healthcare via technology (telehealth) claims saw a staggering increase of over 4,000% during the peak of the pandemic, forcing a decade of digital transformation into mere months. Connected devices allowed high-risk patients to remain safely isolated while ensuring their chronic conditions were still being managed under professional oversight. This period shattered the long-standing skepticism among insurance companies and the healthcare professional fraternity, who finally saw proof that remote, data-driven care was not just possible but often more efficient than in-person visits.
Real-Life Data: The Adherence Revolution
The impact of adding connectivity to injectables is most visible in early success stories. One of the first was the easypod system by Merck KGaA. The easypod demonstrated, in a massive study of over 10,000 patients, that connected systems could maintain very high levels of adherence rates of around 71% over several years. This was a breakthrough in pediatric care.
Today, industry leaders are standardizing these technologies. Research shows that electromechanical devices with digital logs can increase adherence to as high as 90%. For diabetic patients using smart pens like the NovoPen 6 or the InPen, real-time data has proved that even missing two doses in a 14-day window significantly degrades clinical health.
Another connected system is clip-on devices. They represent a simple yet transformative innovation in connected health by turning standard injection tools into smart data-gathering systems. These devices are compact, reusable modules, often shaped like sleeves, caps, or attachments, that snap onto existing disposable autoinjectors or medication pens. Because they are noninvasive, they do not interfere with the internal mechanics of the drug delivery itself; instead, they "observe" the device’s movement from the outside. This allows patients to maintain their familiar treatment routine while adding a digital layer of accountability and support without the high cost of fully integrated electronic hardware.
Once attached, these smart modules use sensors like accelerometers, magnets, or optical scanners to record critical treatment data, including the exact time, date, and dose volume of each injection. They can even track technical details, such as whether the needle was held in place long enough for a full delivery or if the medication was stored at the correct temperature.
This data is stored locally on the clip-on and then wirelessly synced via Bluetooth to a smartphone app. From there, the information is uploaded to a secure cloud, where it can be converted into reports for the patient or shared directly with healthcare providers to monitor adherence and adjust treatment plans in real time.
The Economic Engine: Why Pharma And Insurance Agencies Are Increasingly Supporting Connected Health
While the clinical benefits for patients are clear, the adoption of connected health is also being driven by a powerful economic shift among pharmaceutical companies and health insurers (payers).
For pharmaceutical companies, motivation is twofold: market differentiation and real-world evidence. In a crowded market, a drug paired with a smart injector is more attractive to both doctors and patients than a regular one. Furthermore, connected devices allow pharma companies to collect anonymized, high-quality data on how their drugs perform in the real world, outside the controlled environment of a clinical trial. This data is gold for R&D; helping companies understand why patients might stop treatment or how to optimize dosing for better efficacy.
For insurance companies, the goal is simple: value-based healthcare. Insurers are tired of paying for waste when millions of dollars spent on expensive drugs are flushed down the drain when patients don't follow their schedules.
While technology adds an up-front cost, the true economic value lies in reducing the millions of dollars currently wasted on unused medication and avoidable hospital stays caused by missed or delayed doses.
Challenges: Acceptance And The Regulatory Maze
The path forward is not without friction. Perhaps the greatest hurdle is patient readiness. Many patients, particularly older populations, are hesitant to trade human-centric care for app-centric care. There is a genuine fear of being monitored or failing to use the technology correctly. For connected health to succeed, the technology must be invisible. It should feel like using a standard pen while the data "magic" happens in the background.
As our medical devices become more talkative, a difficult question emerges: Who actually owns the data?
This creates a complex power struggle between patients, pharma companies, and insurers. If a smart pen records that you’ve been skipping doses, should that data belong solely to you and your doctor, or does your insurance provider have a right to see it? Much like black box car insurance, where safe driving leads to lower premiums, the healthcare industry is playing with a premium incentive model.
While this could reward patients for healthy behavior, it also risks creating a digital divide where those who struggle with technology, or those whose bodies simply don't respond to treatment as predicted, are unfairly penalized with higher costs. Navigating these ethical complications will be just as important as the engineering behind the devices themselves.
Another challenge is, as we add microchips, sensors, and lithium batteries to drug delivery devices, we run headfirst into a massive environmental challenge: medical e-waste. Traditional injectables are often single-use plastics, but adding high-tech electronics to a disposable pen is neither economically nor environmentally sustainable. To solve this, the industry is pivoting toward hybrid eco-designs.
Several companies are leading the way with reusable smart bases or clip-ons. In this model, the patient only disposes the needle and drug reservoir, while the expensive connected electronic module snaps onto the next refill. This approach allows the technology to be used for two or three years, dramatically reducing the carbon footprint of digital health and making it a viable long-term solution for a planet already struggling with plastic waste.
Importantly, regulatory complications remain a significant bottleneck. Connected health technologies are often combination products that are part drug, part medical device, and part software. This creates a lack of clarity in how these devices are approved and who is liable if the software glitches. Agencies like FDA and EMA are working to increase awareness and streamline these paths. The FDA’s Digital Health Center of Excellence is now setting stricter standards for cybersecurity and data privacy to build public trust. However, the industry is still navigating a landscape of draft guidances that can delay lifesaving innovations.
The Future: Predictive Intervention
Despite all of these challenges, we are moving one step ahead in connected health, toward a future of predictive intervention. Using AI, future systems won't just record that you missed a dose; they will analyze your behavioral patterns to predict when you are likely to stop your treatment and alert a doctor before a relapse occurs.
The ultimate goal of connected health is the move toward a closed-loop system, where medication is delivered based on real-time physiological needs rather than a static prescription. Imagine a world where a connected injector doesn't just deliver a standard 50 mg dose but instead "talks" to a wearable biosensor that measures drug levels in the blood. If the sensor detects that the previous dose is still active or that the patient’s inflammatory markers are low, the system could suggest a real-time change by adjusting the dose up or down for that specific hour.
We see the early stages of this in the artificial pancreas, i.e., automated insulin delivery systems for diabetes. But the goal is to expand this to other therapeutic areas like oncology. This level of precision ensures that a patient gets exactly what they need — no more, no less — minimizing side effects and maximizing the drug's impact.
Connected health is slowly turning the lonely act of self-injection into a collaborative process. By making the invisible visible, we aren't just improving adherence; we are giving patients the confidence that their therapy is working and giving doctors the tools to save lives long after the clinic doors have closed. At its heart, connected health isn't about replacing doctors with machines; it's about using technology to build a stronger partnership between doctors and patients for better, faster healing.
References:
- Santiago F. Scagliusi, Luis Giménez-Miranda, Pablo Pérez-García, Alberto Olmo-Fernández, Gloria Huertas-Sánchez, Francisco J. Medrano-Ortega, Alberto Yúfera-García. Wearable Devices Based on Bioimpedance Test in Heart-Failure: Design Issues. Rev. Cardiovasc. Med. 2024, 25(9), 320. https://doi.org/10.31083/j.rcm2509320
About The Author:
 Ajay Chintawar is an associate director at Teva Pharmaceuticals with over 17 years of experience in product design, development, regulatory pathways, and manufacturing science and technology. Chintawar’s work primarily focuses on medical devices, drug delivery devices, and combination products. Over the course of his career, he has contributed to the development and commercialization of innovative healthcare products across global markets. Chintawar has previously worked with leading organizations, including Philips, with professional experience spanning India, Europe, and the United States. He brings extensive expertise in bridging engineering, product development, and manufacturing to support the successful delivery of complex healthcare technologies.
Ajay Chintawar is an associate director at Teva Pharmaceuticals with over 17 years of experience in product design, development, regulatory pathways, and manufacturing science and technology. Chintawar’s work primarily focuses on medical devices, drug delivery devices, and combination products. Over the course of his career, he has contributed to the development and commercialization of innovative healthcare products across global markets. Chintawar has previously worked with leading organizations, including Philips, with professional experience spanning India, Europe, and the United States. He brings extensive expertise in bridging engineering, product development, and manufacturing to support the successful delivery of complex healthcare technologies.
