Microneedle Delivery Of Telomerase Protects Skin From Radiation-Induced DNA Damage
By Angela Hu, John P. Cooke, MD, Ph.D., Anahita Mojiri, Ph.D., Houston Methodist Research Institute
For millions of cancer patients, the visible scars of radiotherapy are a daily reminder that lifesaving treatment often comes at a cost. More than half of all cancer patients receive radiotherapy, which relies on ionizing radiation to induce DNA damage and generate reactive oxygen species (ROS) that ultimately eliminate cancerous cells. However, this treatment inevitably harms healthy, visible cutaneous tissue. Approximately 95% of radiotherapy recipients develop radiation-induced skin reactions, such as erythema, dry and wet desquamation, secondary ulceration, and infection. While emollients and corticosteroids are current treatments to alleviate symptoms, there is no definitive solution to prevent or significantly reduce radiation-induced skin injury. To improve patients’ quality of life, there is a critical need for interventions that can protect human skin from radiation-induced damage.
A team at the Houston Methodist Research Institute turned to an unexpected protein: telomerase. The telomerase holoenzyme maintains and extends telomeres at the end of linear chromosomes, thus extending the cellular lifespan and replicative capacity. Its catalytic subunit, telomerase reverse transcriptase (TERT), lengthens telomeres using an intrinsic RNA template. However, the team’s previous works, Mojiri et al. and Qin et al., demonstrated a noncanonical function of TERT: its ability to reduce DNA damage and senescence-associated phenotype factors in prematurely aging cells. The team hypothesized that the pretreatment of TERT mRNA delivered by lipid nanoparticles (LNP) could protect human primary skin cells and human skin ex vivo from irradiation.
And the team was right. Their study revealed that TERT mRNA pretreatment enhanced genomic DNA repair and reduced mitochondrial ROS production and apoptosis in both human skin cells and skin tissue, most likely through an accelerated repair process. Of interest, in cell culture, accelerated DNA damage repair was achieved without altering the length of telomeres.
DNA Damage, Aging, And Senescence: Why They Matter
To understand the broader significance of this study and its findings, it is essential to recognize the interconnected nature of DNA damage, aging, and cellular senescence. Ionizing radiation generates substantial DNA damage and oxidative stress. In response to this stress, cells enter senescence, a stage of permanent growth arrest characterized by the release of senescence-associated secretory phenotype (SASP) factors, leading to inflammation and progressive fibrosis. While SASP factors are initially protective, their accumulation drives chronic inflammation and tissue degradation. In skin, this manifests as impaired wound healing and accelerated aging.
This study illuminates how TERT interrupts this destructive cascade. By enhancing DNA repair and reducing mitochondrial oxidative stress, TERT maintains cellular function and prevents cells from transitioning into a senescent, pro-inflammatory state. This would reduce many aging and damage-associated skin phenotypes.
A Noncanonical Function Of TERT: Repair Of Genomic DNA Damage
The team observed that pretreating keratinocytes and dermal microvascular endothelial cells with TERT mRNA reduced both early and late apoptotic rates, while also limiting mitochondrial reactive oxygen species production that typically amplifies apoptosis and tissue injury. Further experiments revealed that TERT-dependent radioprotection is potentially mediated by enhanced homologous recombination, rather than telomere lengthening. In doing so, this work strengthens prior work linking TERT to DNA repair and illuminates an underappreciated role for telomerase in promoting genomic recovery.
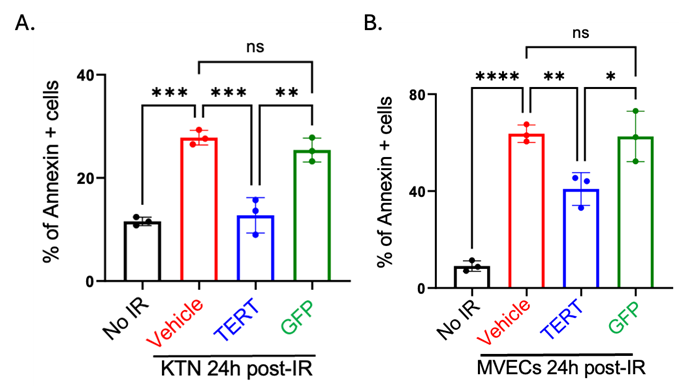
Figure 1. TERT mRNA treatment reduces apoptotic human keratinocytes and dermal microvascular endothelial cells. Annexin V-positive cells indicate apoptotic cells. (A) Quantitative analysis of annexin V-positive cells in indicated groups of human epidermal keratinocytes (KTN). (B) Quantitative analysis of annexin V-positive cells in indicated groups of human dermal microvascular endothelial cells (MVECs). Adapted from Li et al.1
These findings may raise some eyebrows. After all, telomerase is constitutively activated in 80% of cancer cells. However, mRNA telomerase does not integrate into the genome. TERT mRNA is expressed transiently, limiting concerns about uncontrolled proliferation.
A Therapeutic Vision For TERT mRNA
How can TERT be administered in a way that is clinically feasible? To address this, the team developed and optimized a microneedling approach in which TERT mRNA is encapsulated in an ionizable MC3-LNP.
This method builds on the team’s previous study that compared two types of LNPs, ionizable MC3-LNP and cationic DOTAP-LNP, to deliver TERT mRNA to human skin. In this study, they found that the MC3-LNP formulation generated nearly twice as many TERT-positive cells than other delivery vehicles in human skin. Differences in LNP composition may explain the difference in transmission efficacy. Cationic LNPs, like DOTAP, carry a permanent positive charge and contribute to high systemic toxicity. On the other hand, ionizable LNPs remain neutral under physiological conditions and are generally less toxic.
Microneedling was selected as the delivery approach because the intact stratum corneum presents a significant barrier to nanoparticle penetration, limiting the efficiency of topical LNP administration. Intravenous delivery was not pursued, as systemically administered LNPs predominantly accumulate in the liver and do not effectively target cutaneous tissue. Microneedling provides a minimally invasive method to transiently disrupt the epidermal barrier and create microchannels that facilitate localized dermal deposition of nanoparticles. Microneedling is widely used in dermatologic practice and has been shown to enhance intradermal delivery of nucleic acids and biologics through transient microchannel formation without causing permanent tissue disruption.
In this study, ex vivo human skin tissue was microneedled using the Dr. Pen Ultima M8 device, with needle penetration depth set at 2.0 mm to enable delivery into the dermal layer. Following microneedling, the MC3-based LNP formulation encapsulating TERT mRNA was applied, allowing enhanced dermal uptake and localized expression.
After treatment, the skin samples were incubated for 24 hours to allow for TERT expression before being exposed to radiation. The results were intriguing. Pretreatment of human skin reduced radiation-induced DNA damage signal, as indicated by γH2A.X and p53BP1, and senescence markers, as indicated by p21 and p16 (Fig. 2B). TERT-pretreated skin also exhibited significantly fewer apoptotic cells at 24 hours post-irradiation. Crucially, epidermal structure and telomere length were preserved, reinforcing that the protective effects stemmed from enhanced repair mechanisms, not from telomere extension and uncontrolled proliferation.
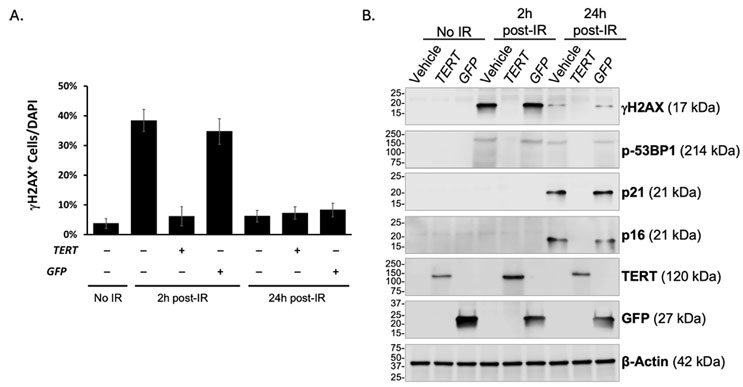
Figure 2. Ex vivo human skin pretreated with TERT mRNA reduced radiation-induced γH2A.X signal and senescence markers. (A) Percentage of cells stained positive for γH2A.X in indicated conditions. (B) Western blot analyses of whole cell lysates collected from non-treated skin samples (lanes 1-3), skin samples harvested 2 hours post irradiation (lanes 4-6), or skin samples harvested 24 hours post irradiation (lanes 7-9). Adapted from Li et al. 1
TERT And mRNA Therapeutics
Taken together, these findings highlight that an mRNA TERT delivery strategy is technically possible and potentially an effective therapy. If translated clinically, this approach could reduce the acute and chronic adverse effects of radiation on human skin. More broadly, this work expands the potential of mRNA technologies and telomerase therapeutics, protecting tissue from adverse injury or aging-associated events.
The team’s full findings were published on January 7, 2026, in Cell Press Molecular Therapy.
References:
- Li, S., Chang, D. F., Court, K. A., Nguyen, T. K. C., Shah, V. V., Morales, E., … Cooke, J. P. (2026). Telomerase mRNA therapy protects human skin against radiation-induced DNA damage. Molecular Therapy, 34(1), 330–347. doi:10.1016/j.ymthe.2025.09.029
About The Authors:
 Angela Hu is a fourth-year undergraduate student at Rice University, studying biosciences with a concentration in cell biology and genetics. She has been working with John P. Cooke, MD, Ph.D. since 2023, researching telomerase-based therapies to mitigate phenotypes associated with cell senescence, vascular aging, and heart failure.
Angela Hu is a fourth-year undergraduate student at Rice University, studying biosciences with a concentration in cell biology and genetics. She has been working with John P. Cooke, MD, Ph.D. since 2023, researching telomerase-based therapies to mitigate phenotypes associated with cell senescence, vascular aging, and heart failure.
 John P. Cooke, MD, Ph.D., is the Joseph C. “Rusty” Walter and Carole Walter Looke Presidential Distinguished Chair, professor, and chair of the Department of Cardiovascular Sciences at Houston Methodist Research Institute. He serves as director of the Center for Cardiovascular Regeneration, medical director of the Center for RNA Therapeutics, chief translational officer of the Houston Methodist Academic Institute, and president of the Society for RNA Therapeutics.
John P. Cooke, MD, Ph.D., is the Joseph C. “Rusty” Walter and Carole Walter Looke Presidential Distinguished Chair, professor, and chair of the Department of Cardiovascular Sciences at Houston Methodist Research Institute. He serves as director of the Center for Cardiovascular Regeneration, medical director of the Center for RNA Therapeutics, chief translational officer of the Houston Methodist Academic Institute, and president of the Society for RNA Therapeutics.
Cooke’s research focuses on mechanisms of vascular cell identity, aging, and RNA therapeutics. His group identified the process of transflammation underlying cell fate transitions and has advanced understanding of telomerase in reversing cellular dysfunction associated with aging. As founding director of the RNA Core, he oversees the development and production of novel RNA constructs for research and therapeutic applications.
 Anahita Mojiri, Ph.D., is an assistant professor in the laboratory of Dr. Cooke in the Department of Cardiovascular Sciences at Houston Methodist Research Institute. Her research focuses on vascular aging and genome stability, using induced pluripotent stem cell–derived vascular cells from children with Hutchinson–Gilford Progeria Syndrome (HGPS) and complementary mouse models.
Anahita Mojiri, Ph.D., is an assistant professor in the laboratory of Dr. Cooke in the Department of Cardiovascular Sciences at Houston Methodist Research Institute. Her research focuses on vascular aging and genome stability, using induced pluripotent stem cell–derived vascular cells from children with Hutchinson–Gilford Progeria Syndrome (HGPS) and complementary mouse models.
Dr. Mojiri found that transient expression of telomerase using mRNA could restore vascular function in HGPS models. The restorative effect on HGPS vascular cells appears to be due in part to non-canonical effects of telomerase to promote genomic DNA repair and reduce cellular senescence. Her work advances the understanding of telomerase as a genome-protective factor beyond its classical role in telomere maintenance.
