Prevascularized Bio-hybrid Organ Platforms Are Redefining Cell Therapy Delivery
By: Arash Memarnejadian, Ph.D., senior research scientist, Sernova Biotherapeutics Pericles Calias, Ph.D., chief development officer & head of R&D, Sernova Biotherapeutics Marylyn Rigby, life science executive, Sernova Biotherapeutics
Cell therapy has entered a phase of increasing clinical maturity, with advances spanning donor-derived transplantation, stem cell-derived products, and gene-edited platforms. Yet across these approaches, one challenge continues to determine whether promising therapies translate into durable clinical outcomes: how and where those cells are delivered.1,2
For metabolically active grafts such as pancreatic islets, loss of function is frequently linked not only to intrinsic cell quality but also to the local implantation environment. Hypoxia, inflammation, and immune-mediated injury can compromise grafts before stable engraftment is achieved, limiting durability.2,3 In this context, long-term function depends as much on the implantation environment as on the cells themselves.
Pancreatic islet transplantation in type 1 diabetes provides a clinically informative model of this challenge. Unlike circulating therapies such as CAR T cells, which distribute systemically, islet grafts must survive, integrate, and function within a defined anatomical site.4 As a result, outcomes reflect interactions between the properties of the cells and the characteristics of the implantation environment.
These observations have prompted a shift in focus from optimizing cell products alone to engineering the in vivo conditions that support their function.5,6 This shift has direct implications for drug delivery strategies. If cell survival, localization, retrievability, and long-term performance are governed by the implantation environment, then delivery is appropriately considered an integral component of the therapeutic mechanism.
Conventional Approaches Still Face The Same Old Bottlenecks
Encapsulation technologies were developed to address these limitations by providing a physical barrier between transplanted cells and the host immune system while permitting the exchange of glucose, oxygen, nutrients, and therapeutic proteins. In practice, however, these systems have encountered a persistent constraint: highly metabolic cells require not only molecular access but delivery at rates sufficient to sustain viability and function over time.2,11,12
That is where many immune-isolating systems have struggled. Barriers that improve immune protection can reduce oxygen and nutrient transport, whereas increasing transport may compromise immune isolation. In addition, foreign body response and peridevice fibrosis can further restrict diffusion and degrade performance.11-13 As a result, a persistent design trade-off between immunoisolation and metabolic support has shaped much of the cell delivery field. For oxygen-sensitive endocrine grafts, this trade-off has proven difficult to resolve within a fully enclosed architecture.11,12
That is where many immune-isolating systems have struggled. Barriers that improve immune protection can reduce oxygen and nutrient transport, whereas increasing transport may compromise immune isolation. In addition, foreign body response and peridevice fibrosis can further restrict diffusion and degrade performance.11-13 As a result, a persistent design trade-off between immunoisolation and metabolic support has shaped much of the cell delivery field. For oxygen-sensitive endocrine grafts, this trade-off has proven difficult to resolve within a fully enclosed architecture.11,12
A Shift In Design Philosophy: From Isolation To Integration
An alternative design paradigm that emphasizes integration rather than isolation has been increasingly explored and validated. Instead of isolating transplanted cells from the host, these approaches aim to introduce them into a controlled, vascularized, and clinically accessible environment.12,14 A central feature of this approach is prevascularization. Rather than placing cells into a poorly vascularized site and relying on host vessel formation, prevascularized strategies establish a supportive tissue environment before cell delivery.14 This staged approach is designed to reduce early hypoxic stress, improve engraftment conditions, and increase the predictability of functional outcomes. Within this framework, delivery systems function as more than passive containers; they actively define the tissue environment in which transplanted cells operate, serving as engineered interfaces between the graft and host.
These three delivery paradigms, uncontrolled delivery, enclosed immunoisolation, and prevascularized integration, are summarized in Figure 1.
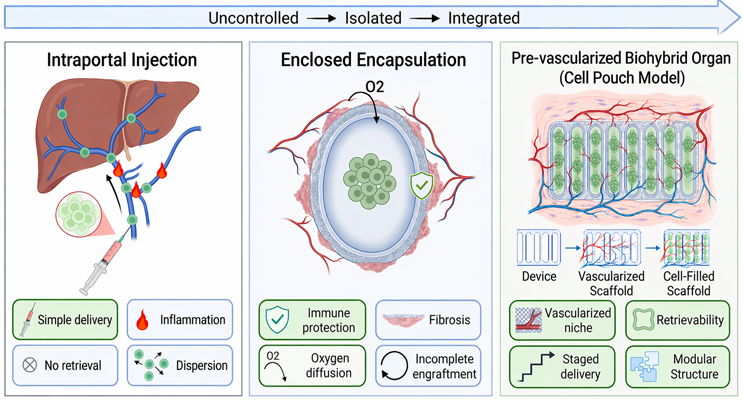
Figure 1. Three conceptual approaches to cell therapy delivery.
(Left) Injection-based delivery results in dispersion, inflammation, and lack of control. (Middle) Encapsulation provides immune protection but induces fibrosis and limits oxygen and nutrient transport. (Right) Prevascularized bio-hybrid organ platforms establish a vascularized niche prior to cell delivery, enabling enhanced engraftment, control, and retrievability. (Created in https://BioRender.com)
The Prevascularized Bio-hybrid Organ Concept
Prevascularized bio-hybrid organ platforms represent a mature implementation of this design approach. The general workflow involves implanting a scaffold, allowing host tissue remodeling and vascularization, and then introducing therapeutic cells into the established vascularized site. This sequence separates two processes that are often forced to occur simultaneously in conventional transplantation: device integration and cell survival.
By establishing a vascularized environment before cell delivery, these platforms modify the initial conditions of engraftment and reduce early physiological stress on transplanted cells. In this context, therapeutic cells are introduced into a defined site with features that support oxygenation and metabolic activity. This design also enables control over cell localization and monitoring while allowing, where applicable, graft retrieval—an advantage that may be difficult to achieve with dispersed delivery approaches.
A Leading Example Of This Shift
One implementation of this design strategy is Sernova Biotherapeutics’ Cell Pouch.15,16 The system is based on a porous scaffold architecture to support host tissue infiltration, angiogenesis, and the formation of vascularized chambers prior to cell loading.
This architecture modifies the biological context of transplantation. Rather than placing cells behind a diffusion-limiting barrier, the platform positions them within a tissue-integrated environment that is designed to permit direct physiological exchange of oxygen, nutrients, glucose, and secreted therapeutic products.15,16
For endocrine applications, this distinction is particularly relevant. Pancreatic islets are sensitive to oxygen availability, and early post-transplant hypoxia has been associated with graft loss.2,3,14 Prevascularized implantation seeks to mitigate this risk by establishing vascular support before cell introduction.
Other device-based strategies reflect the same broader shift toward engineering the implantation environment. ViaCyte’s PEC-Direct/VC-02, for example, was designed as an open, vascularizing delivery device for stem cell-derived pancreatic cells, allowing host integration while relying on systemic immunosuppression.17 In contrast, oxygen-supported macroencapsulation systems such as Beta-O2 have approached the problem from a different angle, seeking to preserve enclosed graft function by supplementing oxygen delivery within a confined architecture.18 Together, these examples underscore that the field is increasingly focused not only on cell source, but also on how device architecture, oxygenation, and host integration shape long-term performance.
Why The Integrated Scaffold Matters
The scaffold is not merely a supporting material; it is the enabling feature of the platform. In functional terms, an integrated porous scaffold enables a set of capabilities that conventional delivery approaches have difficulty combining within a single system:
- It enables host remodeling instead of resisting it. Rather than treating host interaction as purely undesirable, the architecture leverages controlled tissue ingrowth as part of the therapeutic strategy.16
- It establishes vascular support before cell delivery. By preconditioning the implantation site, the platform is intended to mitigate one of the most vulnerable phases in transplantation: the early post-transplant period, when hypoxia-driven graft loss is most pronounced.14,16
- It preserves clinical control. Unlike dispersed injection-based approaches, cells remain localized within a defined and accessible site, supporting monitoring, dose control, and, when necessary, retrieval.16,19
- It creates a modular platform. This is important not only for donor islets but also for stem cell-derived and engineered cell therapies, which are increasingly being developed as standardized and scalable products requiring reproducible delivery environments.16,19
These capabilities align directly with the evolving requirements of modern cell therapies, which demand precise control over dose, localization, and long-term safety, including the ability to intervene or remove the graft if needed.
Just as important, the material and structural design of the scaffold play a central role in biocompatibility and long-term performance. In contrast to enclosed systems that often trigger dense, diffusion-limiting fibrosis, a porous host-integrating architecture can support vascularization while maintaining tissue compatibility over time.15,16,20 This combination of vascular integration and clinical manageability is a key reason the pouch model warrants consideration beyond diabetes alone. Preclinical studies have already extended this concept to a broader range of therapeutic applications, including genetically engineered cells for sustained protein delivery and tissue-based grafts such as thyroid tissue, underscoring its versatility as a controlled, vascularized implantation environment.21-23
What Clinical Experience Is Already Showing
Clinical experience with Sernova’s Cell Pouch in type 1 diabetes is of interest because the platform has demonstrated a set of foundational capabilities that many cell delivery strategies have struggled to achieve in humans. These include implant feasibility, biocompatibility, tissue integration, vascularization, survival of endocrine cells within the device, measurable C-peptide production, and long-term graft residence in a retrievable extrahepatic site.16,24 Evaluation across early-phase clinical studies (ClinicalTrials.gov identifiers: NCT01652911 and NCT03513939) is consistent with a coherent picture of both biological feasibility and the key variables that govern performance.
The first-in-human study demonstrated the ability to generate a vascularized and biocompatible implantation environment, with histologic evidence of viable endocrine tissue within the device.16 Results from the ongoing second study suggest a distinct functional profile compared to intraportal transplantation. Rather than immediate function driven by hepatic exposure, pouch–based islet transplantation follows a more gradual trajectory, consistent with progressive vascular integration and maturation within the engineered niche.24
This pattern is consistent with a central feature of the approach: vascularization is established deliberately before therapeutic cell loading, rather than emerging only after implantation. Clinical experience has also clarified the primary levers that drive outcomes.
Dose matters. Device capacity and islet distribution matter. Immune response management matters.
Data from the later-stage cohorts indicate that improved loading strategies and more effective immune control are associated with enhanced biologic and functional outcomes. At the same time, long-term explant findings showing viable, functional islets up to five years post-transplant provide strong evidence of durable graft survival within the device environment.24
Taken together, these results extend beyond initial clinical validation; they provide a foundation for further translation. The remaining challenges do not appear to stem from a fundamental limitation of the platform architecture, but from continued refinement of cell dose, delivery configuration, and immunological management. For development teams and market observers, that distinction is critical. It separates platforms constrained by biological incompatibility from those that have established in vivo feasibility, long-term compatibility, and durable graft residence—platforms that can now advance through iterative optimization of clinical strategy rather than reinvention of the underlying technology.
The Importance Of This Type Of Platform In The Next Phase Of Cell Therapy
The case for prevascularized bio-hybrid organ platforms becomes even more compelling as the field moves toward stem cell-derived and engineered allogeneic products.25,26 While these approaches address critical supply constraints, they do not eliminate the fundamental requirements for oxygenation, localization, retrievability, or safety. If anything, they may increase their relative importance. A standardized, scalable cell source increases the need for a delivery environment that can reliably support function, control risk, and ensure consistent performance across patients. Whether through open vascularizing devices (PEC-Direct), oxygen-supported macroencapsulation (Beta-O2), or staged prevascularized scaffolds (Cell Pouch), a consistent pattern is observed: durable cell therapy outcomes depend as much on engineering the implantation environment as on the cells themselves.
In this context, cell engineering alone is not sufficient. A gene-edited or stem cell-derived product may reduce dependence on donor tissue, but it still requires a well-controlled biological setting to function predictably and safely. A retrievable, confined, and vascularized platform may provide an additional layer of control, enabling monitoring, intervention, and risk management in ways that cell engineering alone cannot.27
That is why delivery platform and cell engineering should not be framed as competing solutions. The more realistic future is convergence: scalable cell sources paired with implant environments specifically designed to support their function and manage risk.
What Drug Delivery Teams Should Take From This Now
For companies building cell therapy programs, several practical implications follow. Do not treat delivery as a downstream formulation problem. It represents an important determinant of therapeutic success. Build site biology, oxygenation, scaffold design, and retrievability into the platform strategy early. Do not assume immune isolation is the default gold standard. For some highly metabolic grafts, a vascularized and clinically controlled environment may offer a better translational path. Design around the first weeks after transplantation, because early graft loss remains one of the most expensive and underappreciated points of failure in cell therapy.
Finally, think beyond “device” language alone. The most promising systems are not passive containers but integrated bio-hybrid organ platforms: combinations of biomaterials, engineered tissue environments, therapeutic cells, and clinical workflows designed to function as a single, coordinated therapeutic architecture.
Conclusion
The competitive landscape in cell therapy is shifting from who can produce cells to who can make them function reliably and durably in patients. That shift favors delivery platforms that address not only cell placement but also oxygenation, engraftment, safety, and long-term manageability. Multiple platform concepts are now being explored, from enclosed oxygen-supported systems to open vascularizing devices and staged prevascularized scaffolds, with a common lesson emerging: durable outcomes depend on creating a biologically supportive implantation environment. The next generation of durable cell therapies may not come from better cells alone. It may come from building the right biological home for them.
References
- Cheng, A. Y. & Garcia, A. J. Engineering the matrix microenvironment for cell delivery and engraftment for tissue repair. Curr Opin Biotechnol 24, 864-871 (2013). https://doi.org/10.1016/j.copbio.2013.04.005
- Tomei, A. A. Engineering Confined and Prevascularized Sites for Islet Transplantation. Transplantation 102, 1793-1794 (2018). https://doi.org/10.1097/TP.0000000000002290
- Linn, T. et al. Ischaemia is linked to inflammation and induction of angiogenesis in pancreatic islets. Clin Exp Immunol 144, 179-187 (2006). https://doi.org/10.1111/j.1365-2249.2006.03066.x
- Grattoni, A. et al. Harnessing cellular therapeutics for type 1 diabetes mellitus: progress, challenges, and the road ahead. Nat Rev Endocrinol 21, 14-30 (2025). https://doi.org/10.1038/s41574-024-01029-0
- Ho, B. X., Teo, A. K. K. & Ng, N. H. J. Innovations in bio-engineering and cell-based approaches to address immunological challenges in islet transplantation. Front Immunol 15, 1375177 (2024). https://doi.org/10.3389/fimmu.2024.1375177
- Doherty, D. T., Khambalia, H. A., van Dellen, D., Jennings, R. E. & Piper Hanley, K. Unlocking the post-transplant microenvironment for successful islet function and survival. Front Endocrinol (Lausanne) 14, 1250126 (2023). https://doi.org/10.3389/fendo.2023.1250126
- Shapiro, A. M. et al. Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N Engl J Med 343, 230-238 (2000). https://doi.org/10.1056/NEJM200007273430401
- Shapiro, A. M. et al. International trial of the Edmonton protocol for islet transplantation. N Engl J Med 355, 1318-1330 (2006). https://doi.org/10.1056/NEJMoa061267
- Van Der Windt, D. J., Bottino, R., Casu, A., Campanile, N. & Cooper, D. K. Rapid loss of intraportally transplanted islets: an overview of pathophysiology and preventive strategies. Xenotransplantation 14, 288-297 (2007). https://doi.org/10.1111/j.1399-3089.2007.00419.x
- Van Der Windt, D. J., Echeverri, G. J., Ijzermans, J. N. M. & Cooper, D. K. C. The Choice of Anatomical Site for Islet Transplantation. Cell Transplant 17, 1005-1014 (2008). https://doi.org/10.3727/096368908786991515
- Henry, R. R. et al. Initial Clinical Evaluation of VC-01TM Combination Product—A Stem Cell–Derived Islet Replacement for Type 1 Diabetes (T1D). Diabetes (2018).
- O'Sullivan, E. S., Vegas, A., Anderson, D. G. & Weir, G. C. Islets transplanted in immunoisolation devices: a review of the progress and the challenges that remain. Endocr Rev 32, 827-844 (2011). https://doi.org/10.1210/er.2010-0026
- Ludwig, B. et al. Transplantation of human islets without immunosuppression. Proc Natl Acad Sci U S A 110, 19054-19058 (2013). https://doi.org/10.1073/pnas.1317561110
- Pepper, A. R. et al. A prevascularized subcutaneous device-less site for islet and cellular transplantation. Nat Biotechnol 33, 518-523 (2015). https://doi.org/10.1038/nbt.3211
- Kriz, J. et al. A novel technique for the transplantation of pancreatic islets within a vascularized device into the greater omentum to achieve insulin independence. Am J Surg 203, 793-797 (2012). https://doi.org/10.1016/j.amjsurg.2011.02.009
- Gala-Lopez, B. L. et al. Subcutaneous clinical islet transplantation in a prevascularized subcutaneous pouch – preliminary experience. CellR4 4, e2055 (2016).
- Keymeulen, B. et al. Encapsulated stem cell-derived beta cells exert glucose control in patients with type 1 diabetes. Nat Biotechnol 42, 1507-1514 (2024). https://doi.org/10.1038/s41587-023-02055-5
- Ludwig, B. et al. A novel device for islet transplantation providing immune protection and oxygen supply. Horm Metab Res 42, 918-922 (2010). https://doi.org/10.1055/s-0030-1267916
- Holtzinger, A. et al. in 4th IPITA/HSCI/JDRF Summit on Stem Cell Derived Beta Cells (International Pancreas and Islet Transplant Association (IPITA), Cambridge, MA, USA, 2023).
- Pepper, A. R. et al. Diabetes Is Reversed in a Murine Model by Marginal Mass Syngeneic Islet Transplantation Using a Subcutaneous Cell Pouch Device. Transplantation 99, 2294-2300 (2015). https://doi.org/10.1097/TP.0000000000000864
- Memarnejadian, A. et al. Enhancing post-operative hypothyroidism treatment: rat thyroid autotransplantation into a pre-vascularized, retrievable cell pouch device. Front Endocrinol (Lausanne) 16, 1642916 (2025). https://doi.org/10.3389/fendo.2025.1642916
- Olgasi, C. et al. Efficient and safe correction of hemophilia A by lentiviral vector-transduced BOECs in an implantable device. Mol Ther Methods Clin Dev 23, 551-566 (2021). https://doi.org/10.1016/j.omtm.2021.10.015
- Wiseman, S. M. et al. Subcutaneous transplantation of human thyroid tissue into a pre-vascularized Cell Pouch device in a Mus musculus model: Evidence of viability and function for thyroid transplantation. PLoS One 17, e0262345 (2022). https://doi.org/10.1371/journal.pone.0262345
- Witkowski, P. et al. in Diabetologia Vol. 67 222 (Madrid, Spain, 2024).
- Deuse, T. et al. Hypoimmunogenic derivatives of induced pluripotent stem cells evade immune rejection in fully immunocompetent allogeneic recipients. Nat Biotechnol 37, 252-258 (2019). https://doi.org/10.1038/s41587-019-0016-3
- Hu, X. et al. Hypoimmune induced pluripotent stem cells survive long term in fully immunocompetent, allogeneic rhesus macaques. Nat Biotechnol 42, 413-423 (2024). https://doi.org/10.1038/s41587-023-01784-x
- Vlahos, A. E., Cober, N. & Sefton, M. V. Modular tissue engineering for the vascularization of subcutaneously transplanted pancreatic islets. Proc Natl Acad Sci U S A 114, 9337-9342 (2017). https://doi.org/10.1073/pnas.1619216114
About The Authors
 Arash Memarnejadian, Ph.D., is a senior research scientist specializing in cell therapy delivery and bio-hybrid organ technologies. His research focuses on developing implantable platforms that support vascularization, immune management, and long-term function of therapeutic cells. He has been involved in preclinical and clinical programs in islet transplantation and has contributed to advancing bio-hybrid organ approaches for endocrine and protein replacement therapies. Memarnejadian has published in the fields of transplantation, immunology, and regenerative medicine, with a focus on bridging device design and clinical translation. He can be contacted through LinkedIn at: www.linkedin.com/in/arash-memarnejadianian-34855444.
Arash Memarnejadian, Ph.D., is a senior research scientist specializing in cell therapy delivery and bio-hybrid organ technologies. His research focuses on developing implantable platforms that support vascularization, immune management, and long-term function of therapeutic cells. He has been involved in preclinical and clinical programs in islet transplantation and has contributed to advancing bio-hybrid organ approaches for endocrine and protein replacement therapies. Memarnejadian has published in the fields of transplantation, immunology, and regenerative medicine, with a focus on bridging device design and clinical translation. He can be contacted through LinkedIn at: www.linkedin.com/in/arash-memarnejadianian-34855444.
 Pericles Calias, Ph.D. has over 25 years of biopharma industry experience with a strong track record in compound development, pipeline progression, and building research and development organizations. He is currently cofounder of Educational Trainers & Consultants (ETC). Previously, he served as chief operations and development officer at Revolo Biotherapeutics, chief scientific officer at Cerecor, and chief vice president of Global CMC and Product Development at Sucampo Pharmaceuticals. Earlier in his career, Calias held a variety of research and development leadership roles across clinical and nonclinical functions at Shire HGT and Genzyme.
Pericles Calias, Ph.D. has over 25 years of biopharma industry experience with a strong track record in compound development, pipeline progression, and building research and development organizations. He is currently cofounder of Educational Trainers & Consultants (ETC). Previously, he served as chief operations and development officer at Revolo Biotherapeutics, chief scientific officer at Cerecor, and chief vice president of Global CMC and Product Development at Sucampo Pharmaceuticals. Earlier in his career, Calias held a variety of research and development leadership roles across clinical and nonclinical functions at Shire HGT and Genzyme.
 Marylyn Rigby is a life sciences executive with more than two decades of experience across pharmaceutical, biotechnology, and medical technology organizations. Her work has included senior leadership roles supporting corporate strategy, product development, and cross-functional execution in emerging therapeutic areas. She has contributed to initiatives involving biotechnology, medical technology, and patient-centered innovation. Her perspective is also informed by her close personal connection to type 1 diabetes and her interest in technology-driven approaches designed to improve long-term outcomes for patients.
Marylyn Rigby is a life sciences executive with more than two decades of experience across pharmaceutical, biotechnology, and medical technology organizations. Her work has included senior leadership roles supporting corporate strategy, product development, and cross-functional execution in emerging therapeutic areas. She has contributed to initiatives involving biotechnology, medical technology, and patient-centered innovation. Her perspective is also informed by her close personal connection to type 1 diabetes and her interest in technology-driven approaches designed to improve long-term outcomes for patients.
